
The first and only oral film formulation of riluzole1
For patients who have difficulty swallowing some medications, EXSERVAN™ may be an option.1-3

One 50-mg oral film twice daily1
- Taken at least 1 hour before or 2 hours after
a meal1 - Rectangular-shaped with a honey
lemon flavor1
Each film is individually packaged in afoil pouch1 - Average time to dissolve: 2 minutes2

Administering EXSERVAN™
Advise patients to read the FDA-approved patient labeling (Instructions for Use)1
- The patient should place the film on the top of the tongue, and let it adhere and dissolve. Do not cut or split the film1
- Patients should not drink liquid, chew, spit, or talk while
EXSERVAN™ is dissolving1 - Instruct patients to swallow saliva in a normal manner as
EXSERVAN™ dissolves1
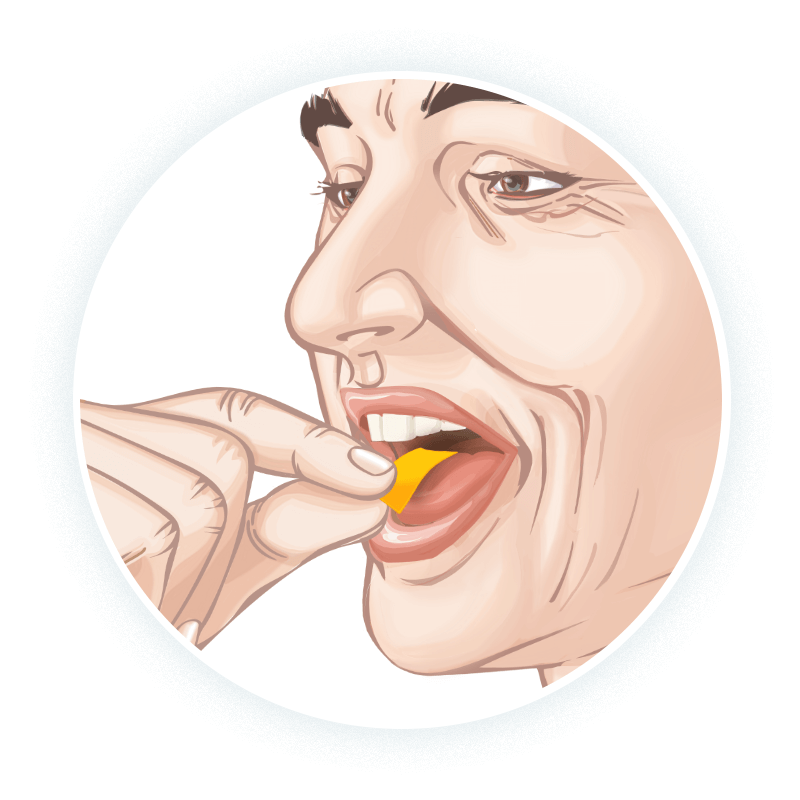
EXSERVAN™ Instructions for Use
Click on each Step below to see full administration instructions.

Patients should make sure their hands are clean and dry before handling

Instruct patients to fold the foil pouch along the solid line at the top. While keeping the top of the pouch folded over at the solid line, patients should tear down at the slit along the arrow on the side of the pouch to open.1
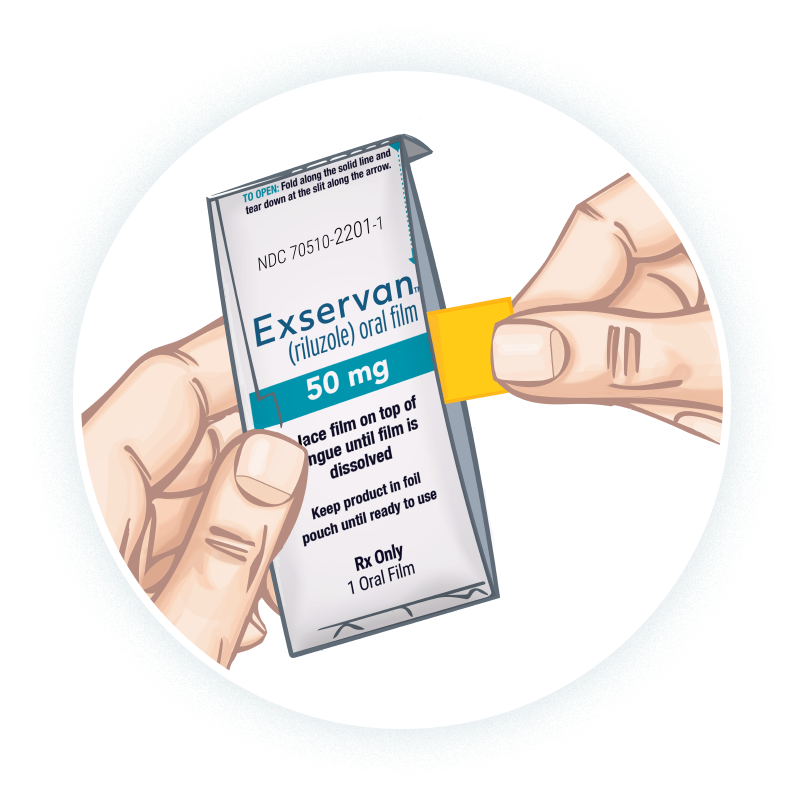
Patients should remove
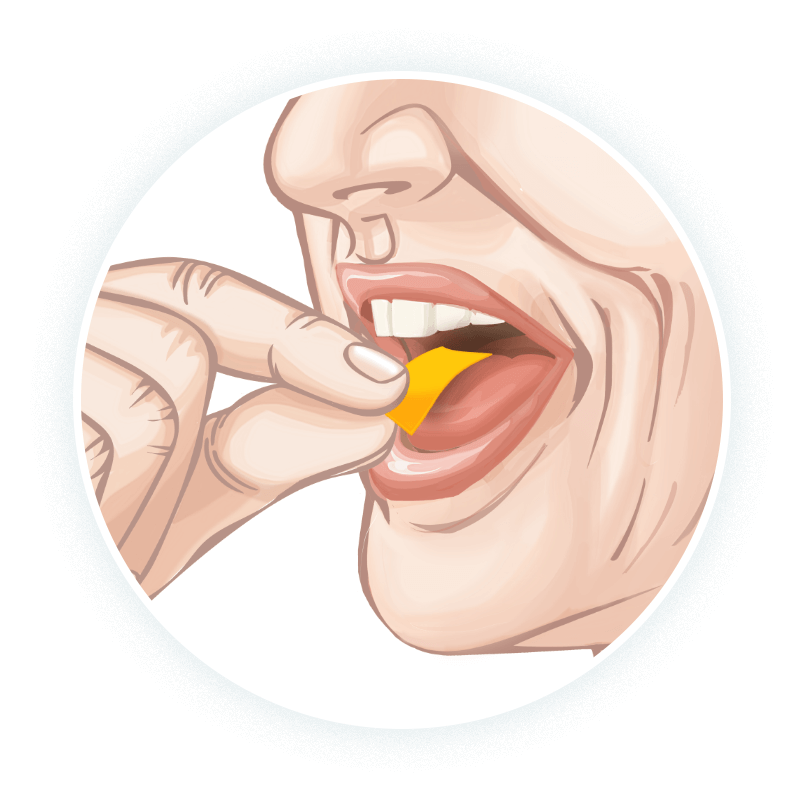
Advise patients to place

The patient should close their mouth and swallow saliva normally as
Mitsubishi Tanabe Pharma America, Inc. is committed to advancing treatment options for people living with ALS.
Discover another treatment option to consider in your patients' ALS journeys
Learn More-
References:
EXSERVAN™ (riluzole) Prescribing Information. Jersey City, NJ: Mitsubishi Tanabe Pharma America, Inc.; 2021.- Data on File. Mitsubishi Tanabe Pharma America, Inc., Jersey City, NJ.
- Center for Drug Evaluation and Research (US). 2017. EXSERVAN (riluzole). Washington DC: US Food and Drug Administration, Center for Drug and Evaluation Research. https://www.accessdata.fda.gov/
drugsatfda_docs/ nda/ 2019/ 212640Orig1s000MedR.pdf. Accessed April 2, 2021. - RILUTEK® (riluzole) Prescribing Information. Cary, NC: Covis Pharmaceuticals Inc.; 2016.